We've learned how constructive and destructive interference of atomic orbitals explains the formation of bonding and anti-bonding orbitals. We also leaned about two types of bonding: σ and π bonding. So you might expect that for polyatomic molecules, all you need to do is put the atoms of the molecule near each other in the right geometry and then see what σ or π bonds form between all the atomic orbitals.
Well, it is almost that simple. The only problem is that for most molecular geometries the atomic orbitals on an atom do not point in the right direction for a σ or π bond to form. Let's look at BF3 as an example. From VSEPR we know the geometry around the Boron atom should be trigonal planar.

But for a Boron atom all the valence elelctrons are in the 2s, 2px, 2py, 2pz orbitals. Recall their shapes:
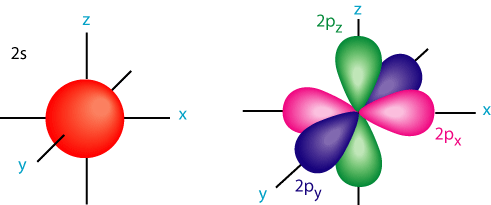
The problem you'll find is that there's no way you can put three Fluorine atoms around the s and p orbitals of Boron in a trigonal planar configuration and form 3 equivalent σ or 3 equivalent π bonds. Yet, we know the B-F bonds are all equivalent because they all have the same bond dissociation energy.
Actually, what happens is that as you bring the three Fluorine atoms near Boron, the atomic orbitals on Boron change (or hybridize) so that they can form σ bonds in a trigonal planar shape.

In this example, one s, and two p orbitals, i.e., px, and py, hybridize to form 3 new orbitals that point along the correct direction to form σ bonds with all 3 Fluorines. This is called sp2 hybridization;
Let's look at another example, BeF2. From the VSEPR model we know its structure is

In Be, the s and px orbitals hybridize to give two similar sp hybrid orbitals.

Remember that the atomic orbitals are standing waves associated with the electrons bound to a nucleus. When you bring atoms together the boundary conditions for these standing waves change and so the standing waves which were the atomic orbitals change. That is all hybridization is. It's analogous to holding down and releasing a violin string while you're playing. There's one standing wave (one frequency) while you're holding down the string, and another standing wave (another frequency) when you release the string.
Let's conside another example, CH4.
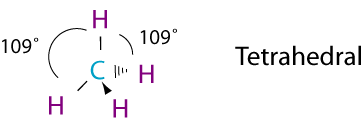
To get these bonds you hybridize one s and three p orbitals. These are called sp3 hybrid orbitals.

Sometimes it is not necessary for all the valence electron orbitals to hybridize. For example, ethylene has the following structure:

The bonds between C and H are all σ bonds between sp2 hybridized C atoms and the s-orbitals of Hydrogen. The double bond between the two C atoms consists of a σ bond (where the electron pair is located between the atoms) and a π bond (where the electron pair occupies the space above and below the σ-bond).

You should remember that we learned about molecules where the central atom gets more than an octet of electrons.

We learned earlier that the extra bonding electron pairs are possible if we include the d-orbitals of phosphorous. This is done by forming hybrid orbitals from s, p, and now d orbitals. For trigonal bipyramidal the central atom is bonded through dsp3 hybrid orbitals.
In the case of molecules with an octahedral arrangement of electron pairs, another d-orbital is used and the hybridization of the central atom is d2sp3
In summary
| Total # of L.P. and B.P about atom | Arrangement | Hybridization |
|---|---|---|
| 2 | linear | sp |
| 3 | trigonal planar | sp2 |
| 4 | tetrahedral | sp3 |
| 5 | trigonal bipyramidal | d sp3 |
| 6 | octahedral | d2sp3 |
What is the hybridization of Xe in XeF4?
Starting with the # of valence electrons = 8 + 4 ( 7 ) = 36 e- (i.e., 18 pairs), and using the VSEPR model we predict an octahedral arrangement of electron pairs about Xe:

Therefore we say that Xe has a d2sp3 hybridization.
For much nicer three-dimensional renderings of all the hybrid orbitals visit Mark Winter's Oritron site .
Homework from Chemisty, The Central Science, 10th Ed.
9.39, 9.41, 9.43, 9.45, 9.51, 9.55

